CBD applications from 7 companies advanced by UK food safety authorities
Cannabis Law Report
MARCH 24, 2023
Hemp Today report A batch of applications for CBD products submitted by a UK trade group has been advanced by food safety authorities.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Cannabis Law Report
MARCH 24, 2023
Hemp Today report A batch of applications for CBD products submitted by a UK trade group has been advanced by food safety authorities.

Cannabis Law Report
JUNE 20, 2021
The Food Safety authority of Ireland (FSAI) has invited the owners and managers of small food businesses to attend a free webinar addressing the legalities of selling cannabidiol (CBD), a chemical compound found in cannabis.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Canna Law Blog
DECEMBER 13, 2021
On December 2, Congress introduced the CBD Product Safety and Standardization Act (the Act ), a bipartisan bill that would establish federal standards for food and beverage products containing hemp-derived cannabidiol (CBD). The post New Federal Bill Would Regulate CBD as a Food Ingredient… But What About the Other Cannabinoids?

Canna Law Blog
OCTOBER 25, 2022
We recently got to ask Kai-Friedrich Niermann of KFN+ Law Office some questions about EU Novel Food authorizations. His answers are a must-read for cannabis food businesses interested in the EU market. What is an EU Novel Food authorization? A novel food catalog is maintained in which the status of a food can be reviewed.

Medicinal Genomics
AUGUST 9, 2021
We continue to see several conflations and misconceptions voiced in the discussion about microbial safety standards for cannabis products. The foundation for these misconceptions is the idea that cannabis testing regulations should mimic the food industry. We have not seen any food on the market that is 20% (weight/volume) antibiotic.

Canna Law Blog
DECEMBER 13, 2021
The CBD Product Safety and Standardization Act (the Act ) indicates that the Feds might regulate CBD as a food ingredient, and that it may lead to regulation of other cannabinoids. If the Feds regulate CBD as a food ingredient, it could seriously change the game (in a good way) for hemp CBD companies.

Cannabis Law Report
APRIL 27, 2023
Hemp Today reports the following Food safety officials in the Czech Republic say they will ban the marketing of products that contain CBD and other cannabinoids from hemp, citing EU regulations and a lack of research on the compounds’ health effects.
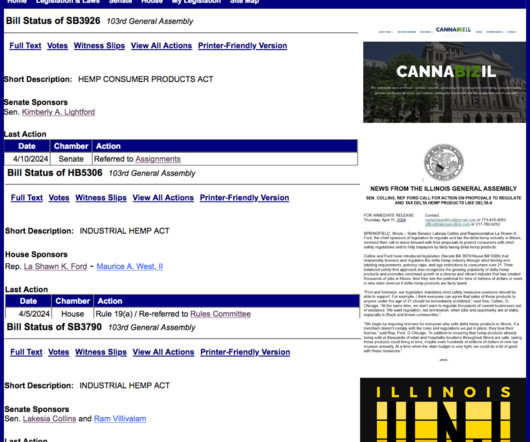
Illinois News Joint
APRIL 11, 2024
Though all these proposals deal with hemp regulations and consumer safety, SB3926 does not align with Ford and Collins’ bills. The bill would create the Intoxicating Hemp-Derived THC Consumer Products Safety Committee, which would include 19 committee members, who would issue a consumer-safety standards report on or before Jan.

Cannabis Law Report
SEPTEMBER 16, 2021
The cannabis plant contains a range of cannabinoids – some of which are psychoactive. Tetrahydrocannabinol (THC) is psychoactive and is considered a narcotic. In Ireland, THC is controlled under the Misuse of Drugs Acts 1977. The affected products are: Source: FSAI. Retailers have been instructed to remove the products from sale.

Cannabis Law Report
OCTOBER 15, 2021
The California Legislature has passed a bill that will allow California consumers to purchase food, beverage, dietary supplements, and cosmetics manufactured with hemp-derived cannabinoids such as cannabidiol (CBD). Some background as to why there is no inter state market for industrial hemp CBD food and beverage products is in order.

Cannabis Law Report
JUNE 3, 2021
introduced legislation on May 21 to ensure hemp-derived cannabidiol (CBD) is regulated by the US Food and Drug Administration (FDA) like other ingredients used in dietary supplements, foods, and beverages. Notably, this restriction only applies to dietary supplements and foods. US Senators Ron Wyden (D-Ore.), Rand Paul (R-Ky.),
Medicinal Genomics
JULY 14, 2022
The truth is that total count tests are not a good indicator of cannabis and hemp product safety for the following reasons: Pathogenic microbes may still be present Testing methods often do not agree Growers are discouraged from using beneficial microbes Growers are encouraged to use remediation. fumigatus, A.

Cannabis Law Report
APRIL 14, 2019
The Spanish Agency for Consumer Affairs, Food Safety and Nutrition (AECOSAN) has struck a blow against hemp food producers in their ongoing efforts to clear a path for hemp extracts in food across Europe. That way of thinking makes extracts, derived from the hemp plant’s leaves and flowers, a “novel” food under EU rules.

Canna Law Blog
JULY 26, 2020
For some background, the European Foods Safety Authority (“EFSA”) previously classified CBD as a “ novel food ” ingredient. A “novel food” is “food that was not used for human consumption to a significant degree within the Union before 15 May 1997, irrespective of the dates of accession of the Member States to the Union.”

Canna Law Blog
OCTOBER 11, 2021
Although DOH launched the Program in November 2020 and began accepting licensing and permit applications for cannabinoid hemp processors, distributors, and retailers, the state agency didn’t initiate the formal rulemaking process until June 2 of this year. Ensuring cannabinoid hemp products meet specific concentration limits.

SpeedWeed
MARCH 21, 2021
As is the case in the United States, Europe lacks sufficiently reliable scientific data to support the safety of products infused with full spectrum hemp extract. and derived products containing cannabinoids, including CBD, as “Novel Food” under the EU Novel Food Catalogue. Read the entire article at The Fresh Toast.

Canna Law Blog
FEBRUARY 21, 2020
Last week, the Food Standard Agency (“ FSA ”), the agency responsible for protecting public health in relation to food in England, Wales and Northern Ireland (collectively, the “UK”), cleared a path for the sale of CBD-infused food for the next 12 months. The EFSA guidance on cannabinoids strongly echoes the U.S.

Cannabis Law Report
JULY 8, 2022
01 Jul 2022 — The UK’s Food and Standard Agency (FSA) has reached a “milestone” after authorizing almost 12,000 cannabinoid (CBD) products to its public list of items that can be marketed in England and Wales. It is an indication that the UK is moving faster than the EU and US at advancing safety regulation.”.

Project CBD
JULY 28, 2023
Scialdone is currently founder and president of BetterChem Consulting , which provides consulting services worldwide in the chemical, food, plant essential oil, and cannabis industries. with unknown pharmacological and safety profiles.” with unknown pharmacological and safety profiles.” Dr. Mark A.

Cannabis Law Report
JANUARY 20, 2022
The Oregon Liquor Control Commission (OLCC) recently finalized new administrative rules on Delta-8 THC and other artificially derived cannabinoids as directed by HB 3000 (2021). Delta-8 THC and other Artificially Derived Cannabinoids. Artificially Derived Cannabinoids Allowed in SOME Situations.

Cannabis Law Report
JULY 28, 2021
On 1 June 2021, the Group announced an agreement with Voisin Consulting SARL (“VC”), a life sciences consultancy comprising over 200 professionals in the UK, US and India, with expertise in medical devices, cannabinoids, neurological disorders and addictions.

Canna Law Blog
MARCH 26, 2020
and derived products containing cannabinoids, including CBD , as a “Novel Food” under the EU Novel Food Catalogue. Indeed, pursuant to Regulation (EU) No 2015/2283 (the “Regulation”), a “novel food” is any food that was not significantly used for human consumption within the European Union before May 15, 1997.

Cannabis Law Report
DECEMBER 12, 2019
The CBD regulatory body for the industry, the Association for the Cannabinoid Industry (ACI), has set a deadline of December 20, 2019, for acknowledgement of engagement from ACI members in the CBD Novel Foods application process. All CBD companies must meet the ACI’s Cannabinoid Quality Charter to become a member.

Canna Law Blog
FEBRUARY 14, 2019
The United States is not the only international actor, however, that is concerned with regulating the sale of CBD products, including CBD-infused foods. The European Food Safety Authority (“ EFSA ”), the European equivalent of the U.S. This also applies to extracts of other plants containing cannabinoids.

Canna Law Blog
MARCH 24, 2019
One of the most frequent questions we receive is whether CBD is a lawful additive in foods. The answer is often no depending on the state in which Hemp CBD food products will be sold. On March 13, 2019, AB-228 got some well-needed amendments which put the law much closer to actually allowing Hemp CBD to be included in food products.

Cannabis Law Report
DECEMBER 18, 2019
According to the latest version of the EU Novel Food Catalogue, EU regulators consider that CBD-containing foods and dietary supplements are classified as novel foods. As a result, CBD-containing foods require an authorisation to be legally placed on the EU market according to Article 6 (2) of the Novel Foods Regulation.

NORML
MAY 15, 2019
NORML has submitted written comments to the US Food and Drug Administration ahead of the agency’s scheduled hearing on the regulation CBD-infused products. percent THC) and products containing cannabinoids derived from hemp from the federal Controlled Substances Act.

Illinois News Joint
OCTOBER 18, 2023
As reported earlier , HB4161 focuses mainly on safety concerns, including permits, licensing, testing, taxes, and a new Hemp Social Equity Fund, but HB4161 also contains amendments that would affect registered Illinois medical cannabis patients and cottage hemp operators as well. Yesterday, House Bill 4161 , introduced by Illinois State Rep.

Cannabis Law Report
JULY 1, 2022
Artificially derived cannabinoid (ADC) products are leaving Oregon grocery stores and convenience stores beginning July 1, 2022. ADCs are most often created by putting hemp-derived CBD through a chemical process to develop new cannabinoid compounds, some of which are intoxicating and some of which are not. Conclusion.

Illinois News Joint
OCTOBER 9, 2023
In addition, HB4161 would: Permit the wholesale and retail sale of cannabinoids, hemp concentrate, or any other intermediate hemp cannabinoid products. Public safety is paramount,” said Ford. Perpetuating the racist war on drugs reflexively will only encourage the black market and hurt public safety. Define terms.

Cannabis Law Report
OCTOBER 29, 2021
OAKLAND – California Attorney General Rob Bonta today issued a consumer alert warning Californians of cannabis-infused edibles that are being packaged and sold as copycat versions of popular food and candy products. KNOW THE HEALTH RISKS: Illegal cannabis products present a risk to public health and safety.

Cannabis Law Report
APRIL 2, 2022
UK Food Standards Agency will now proceed through the safety evaluation process. ” Novel Foods. Novel Foods are ingredients which have not been widely consumed by people in the UK or European Union (EU) before May 1997. This status allows Charlotte’s Web to continue selling its products in the U.K.

Canna Law Blog
AUGUST 8, 2023
Today’s public service announcement is that Congress wants to hear from you on a regulatory fix for cannabidiol (CBD) and other hemp-derived cannabinoid products. The short answer is that Congress has another shot at the Farm Bill, which is the primary agricultural and food policy instrument of the federal government.

Cannabis Law Report
JANUARY 27, 2020
We take a look at where CBD regulation might be going and whether we should be concerned about safety writes Rob Verkerk PhD, scientific and executive director, ANH-Intl. We take a look at where CBD regulation might be going and whether we should be concerned about safety. A new ‘problem’: safety. ? 23 January 2020.

The Joint Blog
APRIL 29, 2020
RSE Technology around the world of edible safety and here are the highlights of the interview: Can you explain the concept of edibles expiring? If the cannabinoid is introduced in an appropriate manner and in a protected format such as S? If the cannabinoid is introduced in an appropriate manner and in a protected format such as S?RSE

Veriheal
JANUARY 18, 2023
The British Food Standards Agency (FSA) has also disqualified approximately 100 CBD products for undisclosed reasons. In 2018, legislation was passed by the British Parliament that effectively legalized cannabinoids like CBD and CBDA. The lack of regulation has raised concerns about consumer safety.

Cannabis Law Report
APRIL 21, 2022
Unfortunately, this appetite combined with a lack of access to regulated marijuana has perpetuated both a robust and unregulated illicit marijuana market, and an unregulated hemp industry where intoxicating hemp-derived cannabinoids like Delta-8 THC, Delta-9 THC, THC-O and HHC are freely sold directly to consumers. If the plant tests above 0.3%

Cannabis Law Report
MARCH 26, 2022
The Centre for Medicinal Cannabis (CMC), the Association for the Cannabinoid Industry (ACI), and First November Group, recently announced the launch of a new wide-ranging commission to review regulation and public policy in the United Kingdom’s (UK) legal cannabis industry called the Hodges Review.

Cannabis Law Report
DECEMBER 14, 2021
(“IGC” or the “Company”) today announced positive primary endpoint data for its Phase 1 clinical trial for IGC-AD1, which is a proprietary cannabinoid-based investigational new drug candidate for patients who have Alzheimer’s disease. Food and Drug Administration (FDA) on December 1, 2021. The average age was 80.9 In addition, 66.7%
Veriheal
AUGUST 9, 2023
Recent research reaffirms earlier studies that claim that certain non-marijuana plants contain similar compounds found in cannabis, often called cannabinoids. Although CBD’s full medical benefits and safety profile are still under review by researchers and appropriate regulators, the compound has successfully scaled several legal roadblocks.

SpeedWeed
SEPTEMBER 16, 2021
While he expressed that he “didn’t want to jump in and all of the sudden have the market drop,” he strongly believes in the medicinal benefits of the cannabinoid and how it can help consumers. For Belushi, the answer is simple. READ MORE: Jim Belushi’s Cannabis Mission. View original article.

The Joint Blog
AUGUST 8, 2023
In recent years, the cannabis industry has witnessed a surge of interest in the therapeutic benefits of cannabinoids. While much attention has been focused on the well-known compounds like CBD and Delta-9-THC, a lesser-known cannabinoid called Delta-8-THC has emerged as a topic of interest.

Veriheal
JUNE 1, 2023
The research authors revealed that hemp seed cake, a by-product from hemp oil extraction, contained low cannabinoids, a healthy source of fiber, and crude protein for cattle and other animals. But regulators are unyielding, expressing skepticism that, if allowed, end consumers may risk exposure to unsafe levels of cannabinoid-laced meat.

The Joint Blog
MARCH 21, 2021
But – the simple truth is that yes, the cannabinoid is legal for use in the UK, so what do you need to know? CBD is one of over 100 cannabinoids, alongside THC, that can be found within the cannabis plant. Novel food status applies to any foods which haven’t been widely consumed by people in the UK or EU prior to May 1997.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content