Why Dr. Sue Sisley Sued the DEA for Stonewalling Cannabis Research
NORML
SEPTEMBER 20, 2019
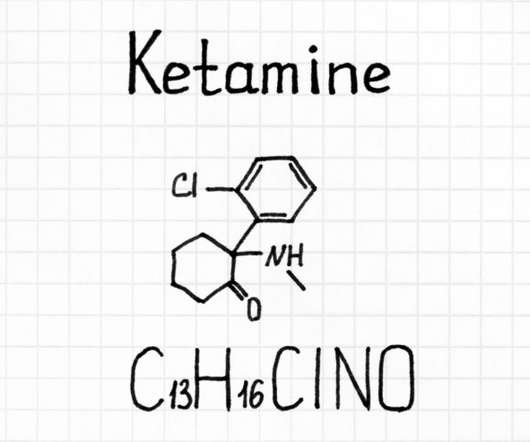
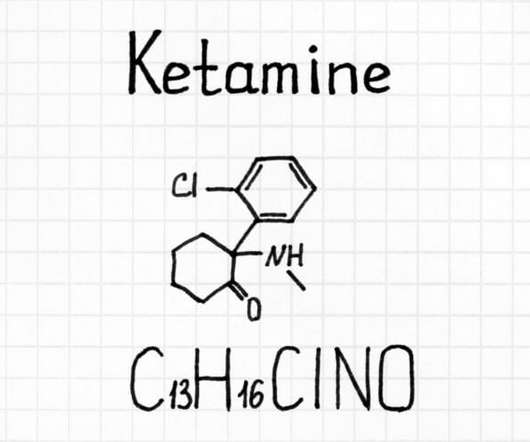
We also spoke with her attorneys, who explained why they believed the DEA broke the law by holding up long-promised medical marijuana research licenses. Essentially, the federal government has monopolized cannabis research, and ElSohly’s product, according to some scientists who’ve seen it, is unsuitable for clinical trials.































Let's personalize your content