Doctor Sues DEA for Right to Give Psilocybin to Ailing Patients
Project CBD
NOVEMBER 8, 2021
Psilocybin, the psychedelic mushroom extract, has been fast-tracked by the FDA to treat depression, but doctors still can’t use it in their practice.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Project CBD
NOVEMBER 8, 2021
Psilocybin, the psychedelic mushroom extract, has been fast-tracked by the FDA to treat depression, but doctors still can’t use it in their practice.

NORML
AUGUST 26, 2019
The US Drug Enforcement Administration (DEA) has once again pledged to take action to better facilitate clinical cannabis research. In 2016, the DEA similarly announced the adoption of new rules to expand to supply of research-grade cannabis, but failed to take any further action.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Veriheal
JANUARY 24, 2023
But recent news of the DEA’s approval of a cocaine derivative for Parkinson’s disease research has left us scratching our heads. After receiving a petition three years prior, the DEA finally answered with action, making plans to deschedule [18F]FP-CIT , a controlled substance derived from cocaine. .”

Canna Law Blog
OCTOBER 17, 2020
The Canna Law Blog has been writing about the Drug Enforcement Agency’s (DEA) interim final rule (IFR) on hemp since its August publication in the Federal Register: Watch Out! The DEA Just Passed a BAD Interim Rule Impacting Hemp CBD and Other Cannabinoids. The DEA Does Not Want You To Worry About Its New Hemp Rule.

Canna Law Blog
OCTOBER 29, 2020
The hemp industry is not the only one that’s pushing back against the Drug Enforcement Administration (DEA)’s hemp Interim Final Rule (the “ Rule ”). If you read this blog, you’ll recall the hemp industry sued the DEA following the release of its Rule in August 2020. 9-THC on a dry weight basis remains controlled in schedule I.”.

Cannabis Law Report
MAY 17, 2021
Rod Kight and Shane Pennington discuss the DEA lawsuit about hemp extracts. As many readers of this blog know, the… Read More.

Canna Law Blog
OCTOBER 29, 2020
The hemp industry is not the only one that’s pushing back against the Drug Enforcement Administration (DEA)’s hemp Interim Final Rule (the “ Rule ”). If you read this blog, you’ll recall the hemp industry sued the DEA following the release of its Rule in August 2020. 9-THC on a dry weight basis remains controlled in schedule I.”.

Canna Law Blog
SEPTEMBER 16, 2020
Unfortunately, the Drug Enforcement Administration (DEA) dropped a surprise interim hemp rule a few weeks ago, which took immediate effect and came as a shock to virtually everyone. The DEA Just Passed a BAD Interim Rule Impacting Hemp CBD and Other Cannabinoids. DEA Interim Final Rule: What Is “Synthetically Derived THC”?
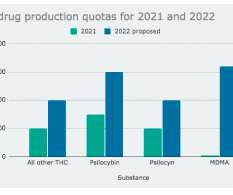
Cannabis Law Report
OCTOBER 15, 2021
The Drug Enforcement Administration (DEA) is proposing a dramatic increase in the legal production of marijuana and psychedelics like psilocybin, LSD, MDMA and DMT to be used in research next year. It wants to double the amount of marijuana extracts, psilocybin and psilocyn, quadruple mescaline and quintuple DMT.

Project CBD
NOVEMBER 8, 2021
Psilocybin, the psychedelic mushroom extract, has been fast-tracked by the FDA to treat depression, but doctors still can’t use it in their practice.

Canna Law Blog
AUGUST 24, 2020
On Friday, the Drug Enforcement Administration (“DEA”) released an Interim Final Rule (the “ Rule ”) that, as we discussed , threatens the hemp industry by treating partially processed hemp extract not intended for consumption (also known as “intermediary hemp”) as a Schedule I controlled substance.

Cannabis Law Report
JULY 23, 2021
“We’re interested in how cannabinoids affect the nervous system, the brain, and in in particular, we’re very interested in Alzheimer’s disease,” said Dr. Tyrell Towle, director of chemistry and extraction at MedPharm. The new DEA research license will now change that.

MJ Business Attorneys
JUNE 29, 2018
Moreover, the ball is in the Drug Enforcement Agency’s (DEA) court. The FDA can make recommendations to the DEA about substances subjected to scheduling under the CSA, but the DEA is the entity with the power to reschedule CBD. In the recent case Hemp Industries Associations v.

Cannabis Law Report
MARCH 24, 2020
The DEA says the proposed rule will see additional registered growers and more diverse variety of marijuana strains available for research. Currently, the DEA has 37 pending applications to grow marijuana for research. DEA Publishes Notice of Proposed Rulemaking On Marijuana Research.

Canna Law Blog
SEPTEMBER 28, 2020
On August 21, the Drug Enforcement Agency (the “DEA”) released an Interim Final Rule (the “ Rule ”), which, in part, suggests that in-process hemp extract shall be treated as a schedule I controlled substance during any point at which its THC concentration exceeds 0.3 percent on a dry weight basis. Emphasis added).

Cannabis Law Report
APRIL 14, 2022
A January letter from the Drug Enforcement Agency (DEA) revealed its official stance that marijuana seeds with a delta-9 tetrahydrocannabinol (THC) concentration lower than 0.3% Shane Pennington, a New York attorney, wrote to DEA requesting the control status of Cannabis sativa L. so long as such parts don’t exceed 0.3% Other guidance.

SpeedWeed
SEPTEMBER 4, 2021
The DEA published a new document in the Federal Register on September 2 requesting an increase in production for certain Schedule I and Schedule II substances so that it can initiate more research studies. . DEA firmly believes in supporting regulated research of schedule I controlled substances. View original article.

Cannabis Law Report
MARCH 21, 2020
“The Drug Enforcement Administration continues to support additional research into marijuana and its components, and we believe registering more growers will advance the scientific and medical research already being conducted,” said DEA Acting Administrator Dhillon. “DEA

Greenspoon Marder Cannabis Law Group Blog
AUGUST 26, 2020
Drug Enforcement Administration (“DEA”) published an interim final rule surrounding hemp and hemp derivatives. Simply stated, when the hemp plant is processed and its cannabinoids are extracted into a crude oil, or similar derivative, concentrated amounts of ? By: Nabil Rodriguez. Last week on Thursday August 20, 2020, the U.S.

Project CBD
JULY 31, 2018
With Congress seemingly poised to legalize the cultivation of industrial hemp for many uses, including the production of biomass for CBD oil extraction, many questions remain unanswered. How will the DEA reschedule CBD now that Epidiolex has been approved as a pharmaceutical CBD isolate?

Cannabis Law Report
AUGUST 26, 2019
DEA Announces Steps Necessary to Improve Access to Marijuana Research. The DEA is providing notice of pending applications from entities applying to be registered to manufacture marijuana for researchers. percent delta-9 THC threshold, is not a controlled substance, and a DEA registration is not required to grow or research it.

Freedom Leaf
OCTOBER 15, 2018
On September 28, the DEA designated Epidiolex —a plant-based CBD pharmaceutical manufactured by the UK-based GW Pharmaceuticals—a Schedule V drug in the government’s list of controlled substances. schedules were created by the Controlled Substances Act (CSA) in 1970 and are interpreted and enforced by the DEA.

The Blunt Truth
OCTOBER 29, 2020
Oregon Senators voice their opposition to the DEA’s hemp rule. New York State released proposed rules for hemp extracts in food, drinks and vapes this week. dea hemp rule. Two weeks ago, we reported that the hemp industry was unhappy with the DEA’s new hemp rule. state ballot roundup. and finally.

Cannabis Law Report
NOVEMBER 22, 2019
MMJ International Holdings, the premier medical cannabis research company, announced that it has received DEA approval to ship THC and CBD from Canada. MMJ International Holdings is developing oral drug products from natural whole plant extract derivatives from the marijuana plant containing THC and CBD.

Canna Law Blog
OCTOBER 3, 2020
That is the DEA’s interim final rule suggesting that in-process hemp extract shall be treated as a schedule I controlled substance during any point at which its THC concentration exceeds 0.3 The DEA Just Passed a BAD Interim Rule Impacting Hemp CBD and Other Cannabinoids. The 2018 Farm Bill Does Not Support the DEA Interim Rule.

Veriheal
DECEMBER 19, 2023
Two methods are employed to process delta-8: one involves the chemical conversion of CBD into delta-8 and delta-9, while the other entails extracting natural delta-8 THC from hemp plants. The Drug Enforcement Administration (DEA) is working on amending the Farm Bill in 2024 to create new rules and regulations regarding synthetic weed.

Canna Law Blog
JUNE 13, 2023
The seeds themselves have little to no THC, regardless of whether they were extracted from or may germinate into a plant that has more than 0.3% On January 6, 2022, DEA issued a letter that responded to a specific query on the treatment of cannabis seeds. trade law and the “reasonable care” standard U.S. federal law.

Cannabis Law Report
JULY 6, 2021
However, CRS notes some observers suggest “a separate, marketable category exists for the plant’s extracted compounds” such as CBD oil. THC cap for lawful hemp products and directed USDA to work with HHS and the DEA on a study of whether that threshold is scientifically backed. Marijuana Moment. Hemp gets $160K in Pa.

The Blunt Truth
AUGUST 27, 2020
The DEA has a new hemp rule. dea hemp rule. The rule makes illegal the production of “wet hemp,” a necessary part of the hemp extraction process that results in a temporary increase in THC. He did not address the DEA’s new rule and its effect on the industry’s survival. And traffic lights go green.

Veriheal
NOVEMBER 6, 2023
In the meantime, the state allows CBD extracts for treating qualifying health conditions with less than 0.9% Despite these strict regulations, in 2014, the state legalized the use of CBD-rich cannabis extracts to treat intractable seizures. The only stipulation is that the extract has less than 0.9%

The Blunt Truth
SEPTEMBER 24, 2020
The DEA faces a lawsuit over their hemp rule. dea regulations. We reported last month that the hemp industry is less than delighted at the DEA’s new rule. They want the court to throw out the rule, claiming that it criminalizes a part of the extraction process. Vermont moves closer to legalized cannabis sales.

Cannabis Law Report
NOVEMBER 16, 2021
As of November 11, 2021, the DEA has yet to issue a binding opinion. Update: Since the posting of this blog, Drug Enforcement Administration officials have made comments suggesting that delta-8 THC is not a federally controlled substance. “Hemp” is defined in the 2018 Farm Bill as the cannabis plant containing no more than.3%

Cannabis Law Report
DECEMBER 15, 2021
The importation of the naturally derived psilocybin into California was completed after the Company’s supply partner Mycrodose Therapeutics was granted an import license from the United States Drug Enforcement Administration (DEA). VANCOUVER, BC , Dec. 13, 2021 /CNW/ – Havn Life Sciences Inc. (CSE:

Canna Law Blog
JULY 26, 2023
Revisit provisions of the Farm Bill or interpretations of the Farm Bill pushed by the DEA, which currently make hemp processors susceptible to civil penalties and felony charges for possession or transport of “hot hemp”, regardless of whether the THC limit is 0.3%, or 1.0%. But while you’re processing that hemp you may be committing felonies!,

Canna Law Blog
JULY 24, 2020
The FDA recommends that those who handle hemp material consult with the Drug and Enforcement Agency (DEA) regarding the control status of such products that are under development. Some manufacturing processes may generate materials, such as intermediates or accumulated by-products, that exceed the 0.3

Canna Law Blog
SEPTEMBER 29, 2023
The Controlled Substance Act prohibits synthetic THCs, and DEA’s 2020 interim final rule stated that any quantity of synthetic THC is controlled. So according to DEA, delta-8 is illegal. This is not the position of DEA, but is evidently the opinion of the aforementioned three-judge Ninth Circuit panel.

Cannabis Law Report
JUNE 12, 2021
Delta-8 THC is derived from CBD extracted hemp plants and has been found to provide a “high” distinct from that experienced from Delta-9, while still falling under the 2018 Farm Bill’s definition of “hemp.”

Cannabis Law Report
MAY 16, 2022
Bravo Botanicals offers a line of organically-certified CBD products made with CBD extracted from their harvest, including cold-pressed drops and creams. Researchers want to force DEA to complete import application evaluation. Welcome to our weekly roundup of CBD and hemp-related legal and regulatory news: CBD. The Detroit News.

Canna Law Blog
DECEMBER 4, 2019
Most of us in the hemp industry are well-aware of the major issues in the interim rules: 15-day pre-harvest testing requirements, total THC, DEA laboratories, and crop insurance to name a few. Moving forward into 2020 many crops would have to be destroyed that have otherwise been able to be used for extraction for the last several years.

Cannabis Law Report
JUNE 29, 2021
Delta 8 is technically a synthetic THC the way most places extract it via an acid wash. Synthetic THC is illegal according to the DEA, so the status of D8 could change anytime as well in TX. That’s about the current state of affairs, but given the two pending decisions, the future of smokable hemp is an unknown.

SpeedWeed
APRIL 27, 2021
Delta-8 is derived from CBD extracted from hemp, but in scientific terms it is delta-8 THC. The demand for delta-8 THC, which is derived from CBD extracted from hemp, is keeping many hemp farmers, processors, and distributors afloat right now. Andrea Golan expects a federal response to delta-8 will first come from the DEA. “We

Cannabis Law Report
JULY 1, 2022
Hemp,” under the Farm Act, was defined as the cannabis plant as well as its “derivatives, extracts, cannabinoids, isomers, acids, salts, and salts of isomers, whether growing or not, with a delta-9 [THC] concentration of not more than 0.3 percent on a dry weight basis.” The Ninth Circuit was not persuaded by either argument.

CannaMD
AUGUST 8, 2020
Drugs, substances, and certain chemicals used to make drugs are classified into five categories (known as schedules ) depending on the Department of Drug Enforcement Agency (DEA)’s definition of the drug’s acceptable medical use and abuse/dependency potential. CBD Scheduling Rule Change. But the government is clear.

Cannabis Law Report
AUGUST 12, 2022
In ruling that delta-8 THC is legal for purposes of trademark protection, the Ninth Circuit opined that products containing delta-8 THC are generally legal because they are hemp derived products and federal law defines hemp as “any part of” the cannabis plant, including “all derivatives, extracts, [and] cannabinoids,” containing less than 0.3%
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content